As an important discipline in quantitative pharmacology, pharmacokinetics focuses on in vivo drug absorption, distribution, metabolism, and excretion to support drug screening, dose design for pharmacodynamic and toxicological experiments, and clinical trial design. Biocytogen's Preclinical Pharmacology Department performs PK assays on antibody drugs, using ELISA and MSD (electrochemiluminescence) platforms, as well as WinNonlin, a professional modeling software for quantitative pharmacology. Biocytogen has adopted FDA-compliant qualitative detection methods and efficient and reliable procedures for blood concentration determination and preliminary PK analysis for monoclonal and bispecific antibodies to help the evaluation of candidate molecules, design of PD experiments, and clinical translational resear



Blood Concentration Analysis of Human (Humanized) Monoclonal Antibodies in Mice
Drug concentration in blood samples is a critical indicator in PK studies, therefore developing an accurate and reliable procedure to detect blood drug concentration is very important. The most frequently used technique for determining antibody concentration is ELISA. Different types of ELISA can be used to analyze monoclonal and bispecific antibody concentrations.

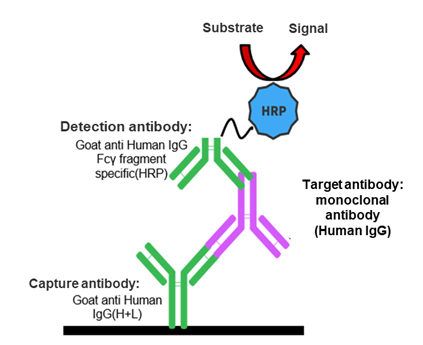
Based on the double antibody sandwich method, an anti-human antibody is used to capture the humanized antibodies in the serum, and then an antibody labeled with HRP that specifically binds to the Fcγ fragment of the humanized antibody is added to detect the drug concentration in the serum.
Features:
The double antibody sandwich ELISA (DAS-ELISA) method can detect the concentrations of subtype monoclonal antibodies of IgG1, IgG2, and IgG4, fusion proteins with intact antibody structures, and bispecific antibodies.
Fixed experimental conditions are available for faster sample testing and more efficient method validation.
The total humanized antibodies in the serum will be tested, including those that bind to soluble targets.
For non-specific antibody binding, it is not able to analyze target antibody concentrations in the case of combination therapy.
This method applies only to the detection of drug concentrations in non-human-derived animal samples.

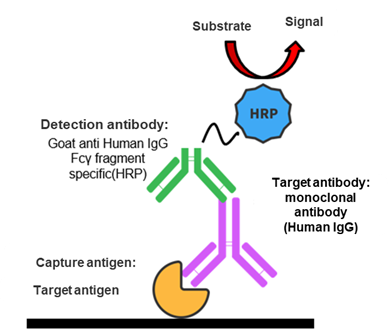
The method is developed based on drug targets. As the target protein of humanized antibody, the capture reagent can specifically bind to the complementarity-determining region (CDR) of the antibody. Then an antibody labeled with HRP that specifically binds to the Fcγ fragment of the humanized antibody is added to detect the drug concentration in the serum.
Features:
The antibody target-based assay is highly specific to the test article and can detect the target antibody in the case of combination therapy.
Methods can be developed, transferred, and thus validated on demand.
Strictly speaking, these methods mainly detect the free humanized antibodies in the serum rather than those already bound to the target.
Blood Concentration Analysis of Human Bispecific Antibodies in Mice

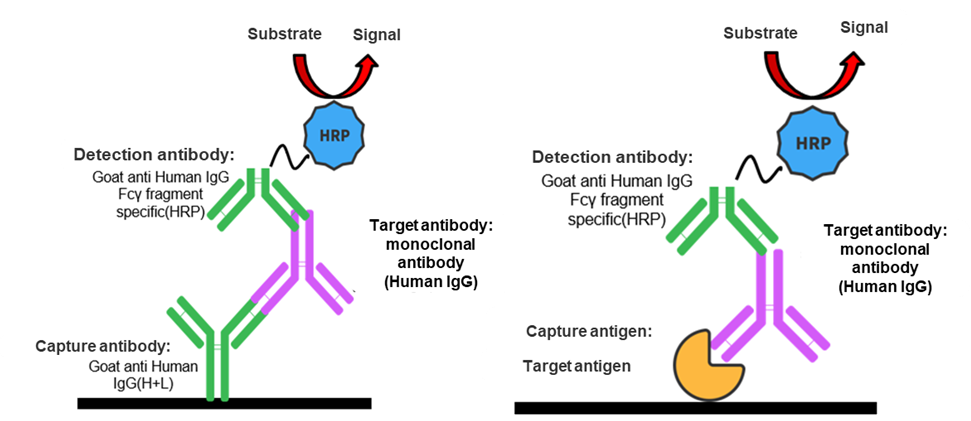
Based on drug targets, the method is developed to detect bispecific antibodies with intact structures in serum. As the target protein of humanized antibody, the capture reagent can specifically bind to the complementarity-determining region (CDR) of the antibody, and the whole detection system will be completed with the addition of another specific target protein (with Biotin or His tags) of bispecific antibodies. Streptavidin-HRP or Anti-His Antibody-HRP is added to generate detection signals.
Features:
The antibody target-based assay is highly specific to the test article and can detect bispecific antibodies with intact structures in serum, as required in preclinical applications.
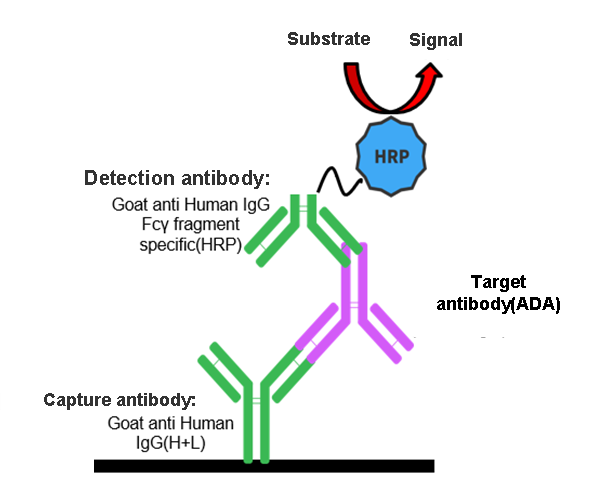
Almost all biological agents may induce anti-drug antibodies (ADAs) to some extent, which may reduce drug efficacy and cause various clinical adverse effects. Antibody drugs can specifically capture ADAs, and ADAs produced in mice can be detected by adding the detection antibody and an HRP-conjugated goat anti-mouse IgG secondary antibody, which specifically binds to the Fc fragment of the antibody in mice.
Features:
ADA assays are commonly used in the industry to detect free ADAs in serum.
The method can qualitatively detect ADAs and obtain preliminary quantitative results based on signal values.
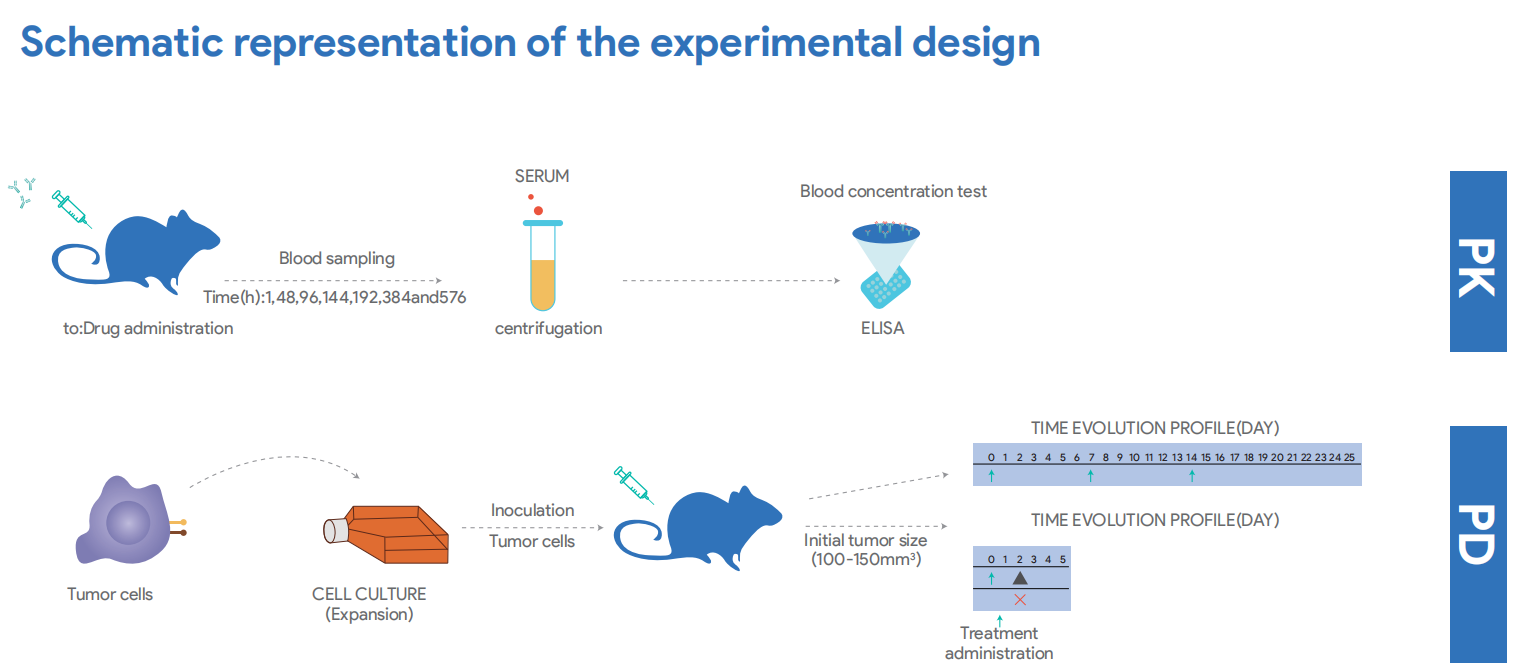
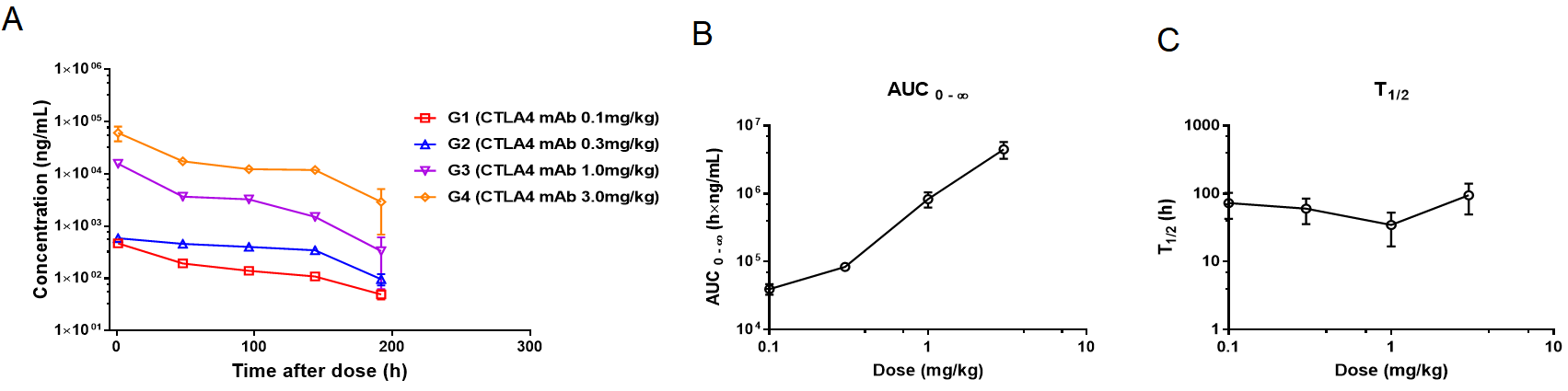
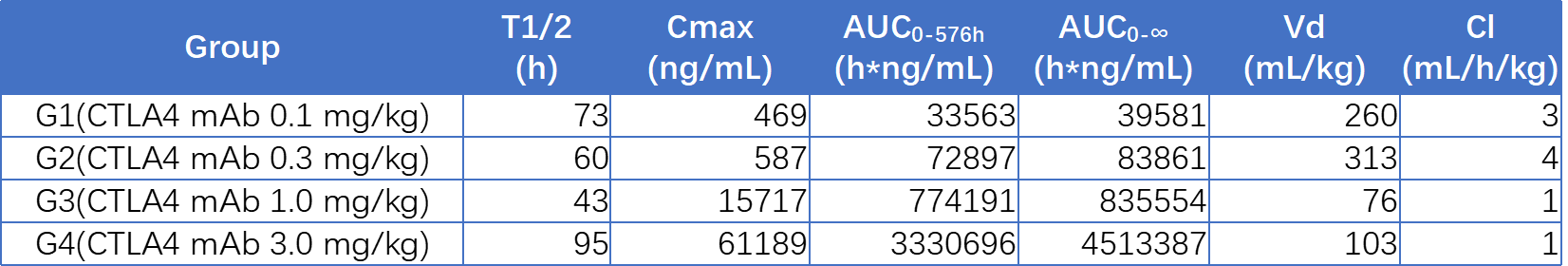
Case: PK Study of Humanized Anti-CTLA4 Monoclonal Antibody






 +86-10-56967680
+86-10-56967680 info@bbctg.com.cn
info@bbctg.com.cn